Pipeline – A
About INTASYL™
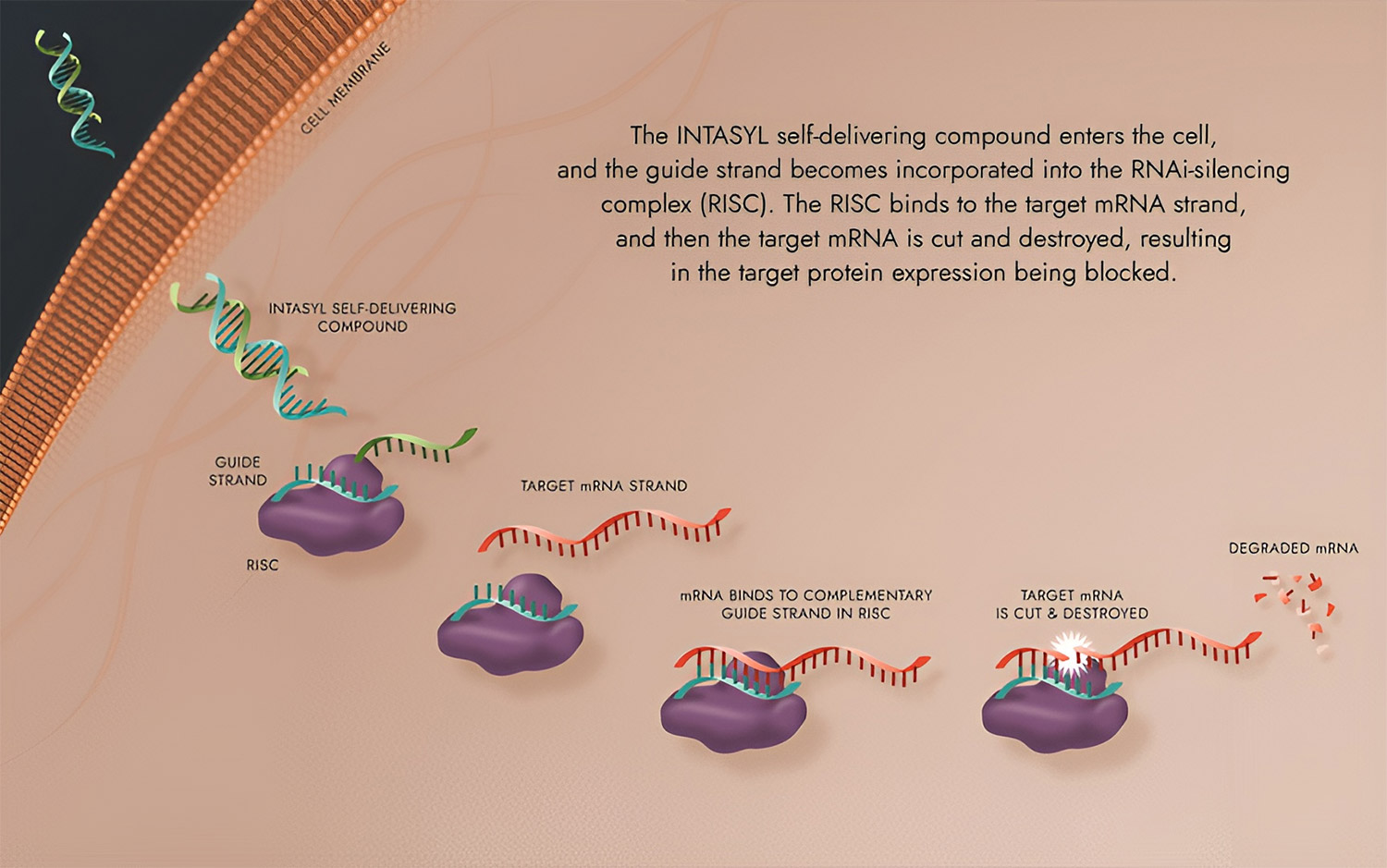
INTASYL ™ is a precision siRNA immunotherapy that targets messenger RNA, which is encoded by genes in the DNA, resulting in the reduction of specific target proteins.
INTASYL is a patented platform from which unique patented compounds are created to silence genes that tumors use to evade the immune system. These self-delivering, chemically modified siRNA compounds can be delivered to a broad range of cell types and tissues without the need for delivery enhancements. INTASYL has a simplified chemical composition that helps reduce toxicity, increase tolerability, and enhance efficacy, and it’s adaptable to both Intra-tumoral and Adoptive Cell Therapy (ACT) applications.
INTASYL compounds have demonstrated activity against multiple gene targets including PD-1, BRD4, CTLA4, TIGIT and CTGF.
Program
Discovery
Preclinical Proof
of Concept
IND Enabling
Studies
IND Clearance
Clinical Phase
PH-762
Read More
PH-762 is an INTASYL compound that reduces the expression of cell death Protein 1 (PD-1), a protein that inhibits T cells’ ability to kill cancer cells.
Target(s) & Indication: PD-1
cSCC, Advanced Melanoma, Merkel Cell
PH-762 is an INTASYL compound that reduces the expression of cell death Protein 1 (PD-1), a protein that inhibits T cells’ ability to kill cancer cells.
Target(s) & Indication: PD-1
Metastatic Melanoma
PH-762
Enhanced TIL Study
(AgonOx Partner)
Read More
PH-762 is an INTASYL compound that reduces the expression of cell death Protein 1 (PD-1), a protein that inhibits T cells’ ability to kill cancer cells.
Target(s) & Indication: PD-1
Metastatic Melanoma, Head and Neck Squamous Cell Carcinoma, and other solid tumors
PH-894
Read More
PH-894 is an INTASYL compound that silences BRD4, a protein that controls gene expression in both T cells and tumor cells, thereby effecting the immune system as well as the tumor.
Target(s) & Indication: BRD-4
Advanced Melanoma, Merkel Cell, HCC, TNBC
Key Clinical Initiatives & Programs
PH-762
- Initiating a dose-escalation study for the treatment of advanced cutaneous squamous cell carcinoma, melanoma or Merkel cell carcinoma.
- Phase 1 clinical trial, PH-762 in combination with Double Positive Tumor Infiltrating Lymphocytes (DP TILS) for the treatment of melanoma and other solid tumors (in partnership with AgonOx).
- For more information about Phio’s clinical trials, contact clinicaltrials@phiopharma.com
PH-894
- Pre-clinical studies have shown this compound has a dual mechanism: the suppression of BRD4 results in T cell activation and makes tumor cells more sensitive to T cell killing.
- Pre-clinical studies of PH-894 show the compound may also have activity for the treatment of viral infections.